Neubauer Counting Chamber
The reference tool for precise manual cell counting in hematology, body fluids, and reproductive analysis
Introduction & Definition
Hemocytometry is the classical laboratory technique used for the manual determination of cell concentration in a known volume of liquid. The method is based on counting cells within a precisely defined grid area and chamber depth, allowing results to be expressed as cells per microliter (µL or mm³).
The Neubauer Counting Chamber, commonly referred to as a hemocytometer, is the standard instrument used for hemocytometry. Its calibrated grid dimensions and fixed chamber depth of 0.1 mm make it a reliable reference method in hematology and body fluid analysis.

Figure 1: Standard Neubauer Counting Chamber with Cover Glass
Specifications & Types
The most commonly used version today is the Improved Neubauer Chamber. The primary difference lies in the central square grid refinement, allowing for more accurate RBC and Platelet counts .
| Feature | Improved Neubauer | Old Neubauer |
|---|---|---|
| Total Area | 9 mm² | 9 mm² |
| Chamber Depth | 0.1 mm | 0.1 mm |
| Large Squares | 9 (1×1 mm each) | 9 (1×1 mm each) |
| Central Square Div. | 25 Small Squares | 16 Small Squares |
| Material | Borosilicate Glass | Standard Optical Glass |
Chamber Design & Key Parts
Hemocytometry depends on strict physical parameters, including uniform chamber depth, precisely etched grid areas, and controlled sample volume. Each structural component of the Neubauer chamber is designed to eliminate volume variation and ensure accurate manual cell counting.
- H-Shaped Moat: Two longitudinal grooves that separate the counting platforms and act as overflow sinks.
- Counting Platforms: Two polished glass surfaces etched with the grid.
- Etched Grid: A 3mm x 3mm grid divided into 9 large squares.
- Cover Glass: A specially ground glass (0.4mm thick) that creates the precise 0.1mm depth.
- Charging Area: The V-shaped slash or the edge where the pipette tip is placed to fill the chamber by capillary action.
- Newton’s Rings: Rainbow-like patterns seen when the cover glass is properly applied, indicating the correct 0.1mm gap.
Grid Sizes & Clinical Uses
In hemocytometry, different grid areas are selected based on cell size, concentration, and distribution. Larger cells with lower concentrations require larger counting areas, while smaller and more numerous cells are counted in subdivided grids.
The grid consists of 9 large squares, each 1mm x 1mm. Total dimensions are 3mm x 3mm.
Corner Squares (WBC)
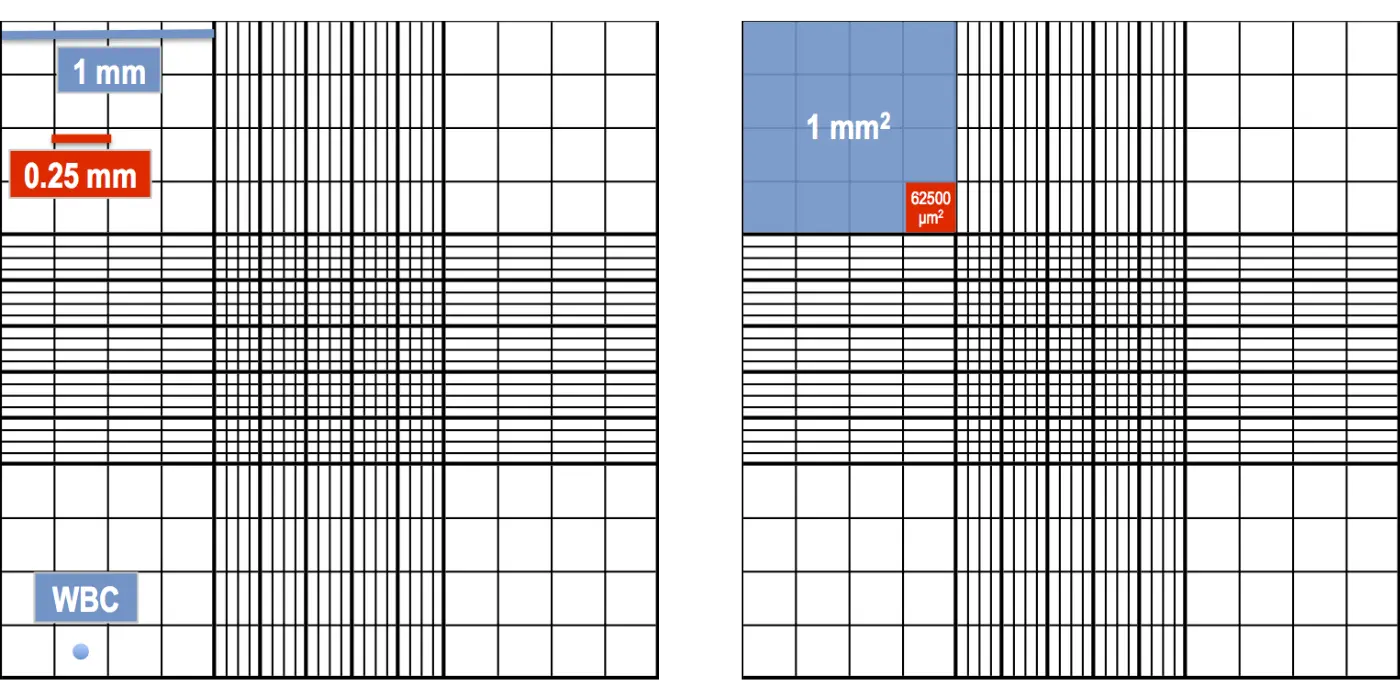
Use: White Blood Cell (WBC) count. These 4 large squares are used because WBCs are larger and less numerous.
Central Square (RBC/PLT)
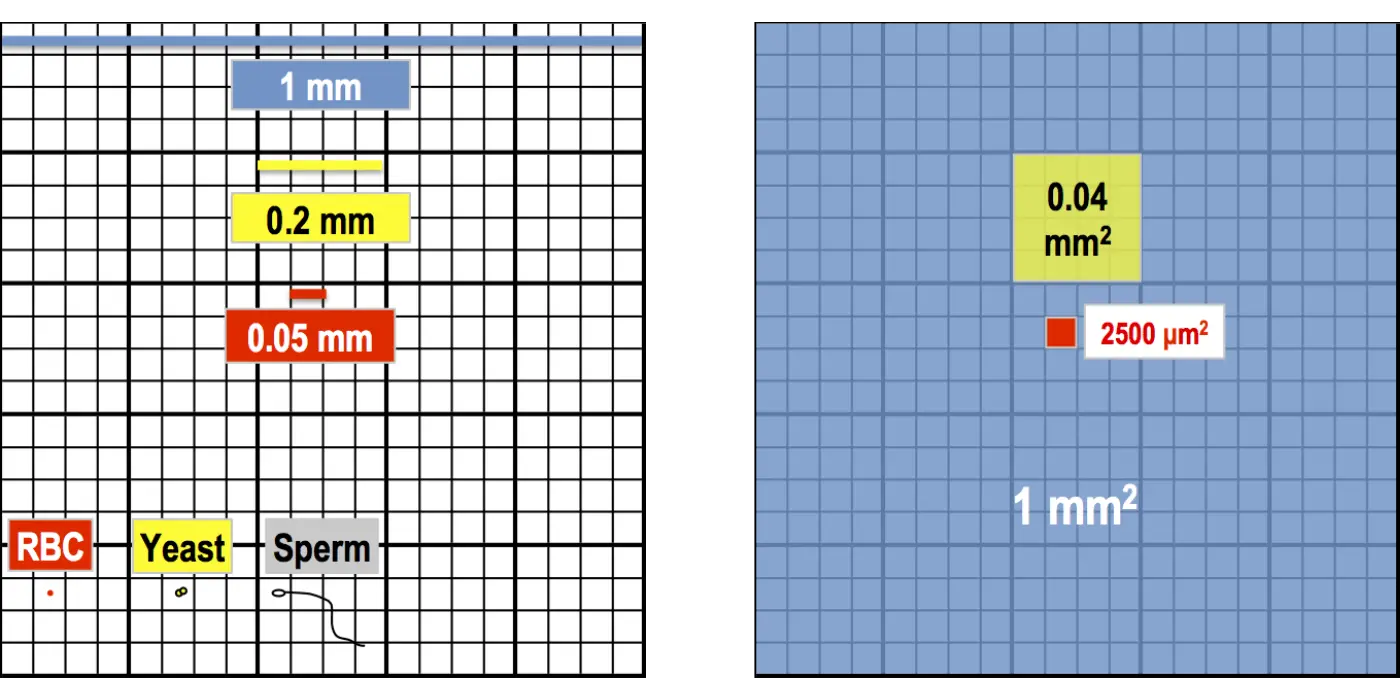
Use: RBCs, Platelets, Sperm, and Yeast cells. The central square is divided into 25 medium squares, which are further subdivided into 16 tiny squares.
Counting Strategies & Rules
Accurate hemocytometry requires standardized counting strategies and strict inclusion–exclusion rules. These rules prevent double counting, reduce observer bias, and ensure reproducibility between duplicate chamber counts.
The 3 Strategies
- The Logical Count: Counting cells in a zig-zag (snake) pattern to avoid double counting.
- The Absolute Count: Counting all cells within the designated grid boundaries.
- The Quick Count: Used for rapid screening when high precision isn’t required.
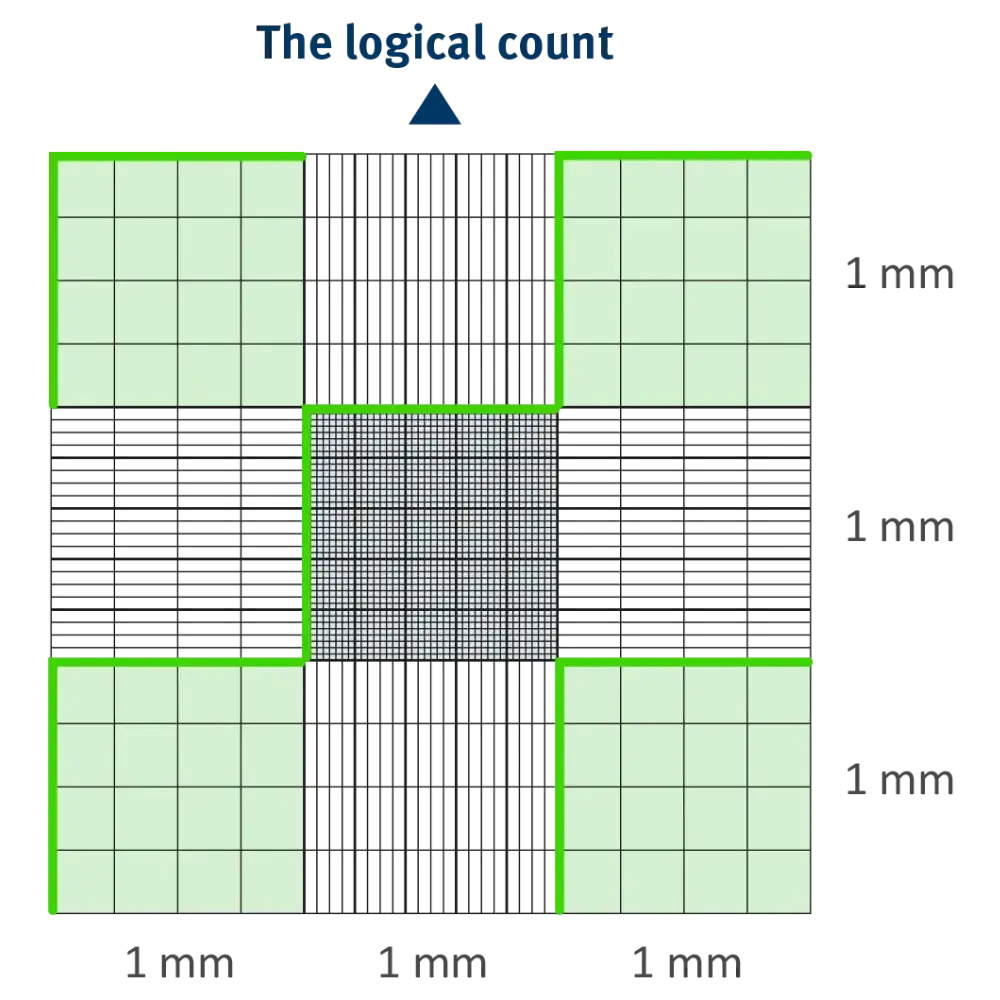
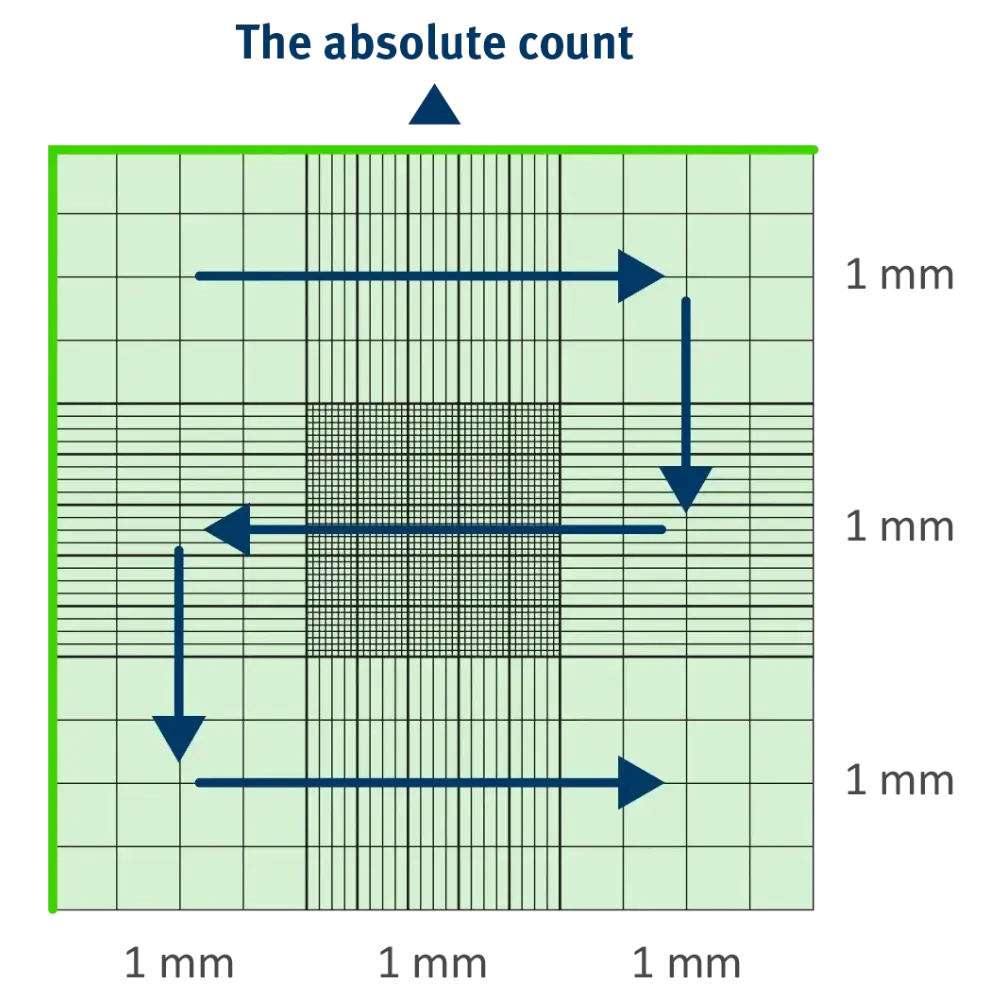
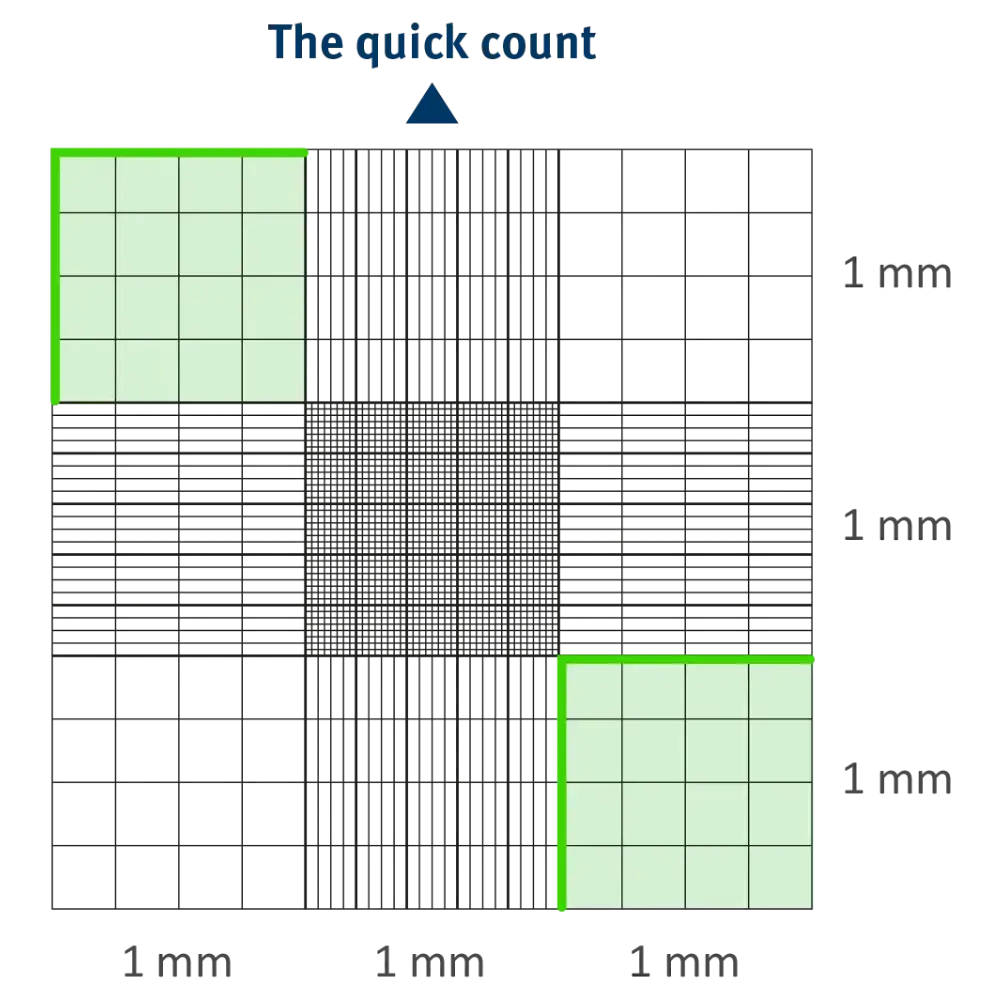
11 Essential Rules for Accuracy
- L-Rule (Inclusion): Count cells on the top and left borders; exclude those on bottom and right borders.
- Cleaning: Always clean the chamber and cover glass with 70% ethanol before use.
- Newton’s Rings: Ensure rainbow patterns appear when pressing the cover glass.
- Sedimentation: Allow cells to settle for 2–3 minutes after charging.
- Overfilling: Never overfill the chamber; the fluid must not flow into the moats.
- Bubbles: If air bubbles are present, clean and recharge.
- Duplicate Count: Count both sides of the chamber; the difference should not exceed 10%.
- Magnification: Use 10x for WBC and 40x for RBC/Platelets.
- Light Intensity: Use low light/closed iris diaphragm to see transparent cells.
- Dilution: Ensure the sample is mixed thoroughly before charging.
- Calculation: Always double-check the dilution factor used.
Procedures & Calculations
Step-by-Step Procedure
All cell counts were performed following the L-rule to prevent double counting of boundary cells.
1. Sample dilution: Prepare 1:20 or 1:200 dilution using the specific diluting fluid.
2. Clean chamber: Wipe with 70% ethanol and a lint-free cloth.
3. Prepare sample: Mix well, dilute if needed.
4. Load chamber: Pipette sample into loading area, filling both sides.
5. Let cells settle: Wait 10-15 minutes in a humidified chamber.
6. Count cells: Use a microscope (10x objective) and apply counting rules.
7. Repeat if needed: Count more samples for accuracy.
8. Clean & store: Clean with ethanol and store safely.
| Cell Type | Diluent | Dilution | Area Counted | Formula Factor |
|---|---|---|---|---|
| WBC | Turk’s Fluid | 1:20 | 4 mm² | 50 |
| RBC | Hayem’s Fluid | 1:200 | 0.2 mm² | 10,000 |
| Platelets | 1% Amm. Oxalate | 1:100 | 1 mm² | 1,000 |
| Sperm | Formalin-Saline | 1:20 | 1 mm² | 200 |
The final step in hemocytometry is calculation of cell concentration. Because the chamber depth and grid area are fixed, the number of cells counted can be mathematically converted into cells per microliter using the dilution factor and the volume examined.
Total Cells (per µL) = (Cells Counted × Dilution Factor) / (Area Counted × Depth)Interactive Cell Calculator
This calculator applies standard hemocytometry principles used with the Neubauer counting chamber to instantly determine cell concentration.
Example (WBC): Counted 160 cells, Dilution 20, Area 4mm², Depth 0.1mm.
Result: (160 * 20) / (4 * 0.1) = 8,000 cells/µL
Frequently Asked Questions (FAQs)
1. What is the depth of the Neubauer Counting Chamber?
The standard depth of an Improved Neubauer Counting Chamber is 0.1 mm. This precise depth is maintained by the cover glass and is crucial for calculating the volume of the fluid being counted.
2. Which squares are used for WBC and RBC counts?
For a WBC count, the 4 large corner squares (1 mm² each) are used. For RBC and Platelet counts, the large central square is used, specifically counting the 5 small squares (4 corner squares and 1 central square within the center grid).
3. What is the “L-Rule” in manual cell counting?
The L-Rule is a counting convention used to avoid double-counting cells. It states that cells touching the top and left (North and West) boundaries of a square should be counted, while cells touching the bottom and right boundaries should be excluded.
4. Why are Newton’s Rings important during chamber preparation?
Newton’s Rings (rainbow-colored interference patterns) appear when the cover glass is perfectly adhered to the chamber supports. Their presence confirms that the chamber depth is exactly 0.1 mm, ensuring the accuracy of the final calculation.
5. How do you clean a Neubauer Chamber?
After use, the chamber and cover glass should be cleaned with 70% Isopropyl Alcohol (Ethanol) and wiped dry with a lint-free tissue (like Kimwipes). Never use abrasive materials as they can scratch the etched grid.
6. Why is manual counting still used instead of automated analyzers?
Manual counting is used as a backup for automated systems, specifically for extremely low cell counts (leukopenia/thrombocytopenia), analyzing body fluids like CSF or Semen, and verifying results when an analyzer flags abnormal morphology.
References:
- Neubauer Chamber (Neubauer Hemocytometer) |Electron Microscopy Sciences | https://www.emsdiasum.com/docs/technical/datasheet/68052-14 – (Accesscd Sep 12, 2024)
- Hemocytometer | Wikipedia | https://en.wikipedia.org/wiki/Hemocytometer – (Accesscd Sep 12, 2024)
- How to count cells with a hemocytometer – ChemoMetec| https://chemometec.com/how-to-count-cells-with-a-hemocytometer – (Accesscd Sep 12, 2024)
- Hemocytometer | https://www.hemocytometer.org | – (Accesscd Sep 12, 2024)
- Vembadi A, Menachery A, Qasaimeh MA.: Cell Cytometry: Review and Perspective on Biotechnological Advances. Front Bioeng Biotechnol. 2019;7:147.
- Electron Microscopy Sciences: Neubauer Haemocytometry.
- Stoddart MJ.: Cell viability assays: Introduction. Methods Mol Biol. 2011;740:1-6




One Comment