‘Revolutionary’ Blood Test Detects 50+ Cancers — But Experts Urge Caution
New data from a large clinical trial show that a single blood test can detect more than 50 types of cancer — including many for which there is no current screening. The results could reshape early-detection strategies — but limitations remain, and real-world impact on mortality is still unknown.
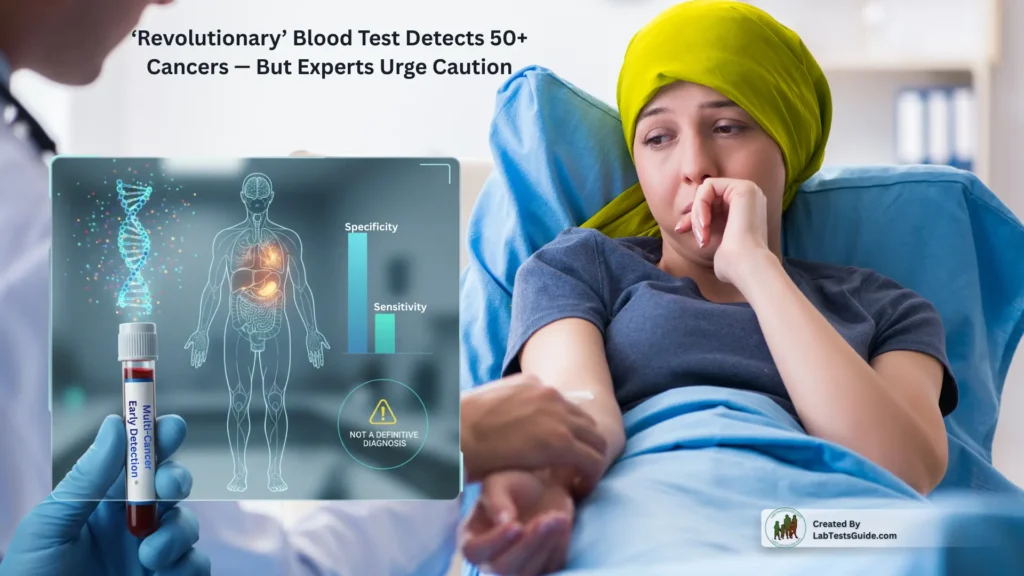
A potentially transformative new tool in the fight against cancer has emerged. According to recently released results from multi-cancer blood test a simple blood draw may help detect over 50 different types of cancer, many of which currently lack any routine screening.
What the New Study Shows
- The data come from the latest phase of the clinical trial known as PATHFINDER 2, which enrolled 35,878 adults aged 50 and older from the United States and Canada with no prior suspicion of cancer.
- Among the 23,161 participants whose results were analyzable after 12 months, a “cancer signal” was detected in 216 individuals (about 0.93%).
- Of those who tested positive, 133 were subsequently diagnosed with cancer — a confirmed-cancer rate of 0.57% overall and a positive predictive value (PPV) of 61.6%.
- Furthermore, around three-quarters of the cancers discovered by Galleri do not currently have standard-of-care screening options — for example, cancers of the pancreas, liver, and ovaries.
- When added to standard screening (for breast, cervical, colorectal, and lung cancers), Galleri increased the number of cancers detected more than seven-fold within a year.
The blood test analyzes cell-free DNA (cfDNA) circulating in the blood — fragments shed by tumour cells — and uses patterns (e.g., DNA methylation) to identify a “cancer fingerprint,” and even predicts the most likely organ or tissue origin of the cancer signal.
Why This Is Promising
- Broader coverage: The test targets dozens of cancer types — including many that today have no screening program. This could substantially expand early detection reach beyond the few cancers routinely screened now.
- Earlier detection: Detecting cancer at early stages improves treatment options and survival chances. The fact that more than half of detected cancers were early stage suggests the test could shift diagnosis to a more treatable phase.
- High specificity, low false positives: The study reports a specificity of 99.6% — meaning only about 0.4% of people without cancer received a “cancer signal detected” result.
- Guiding follow-up testing: The predicted “cancer signal origin” from Galleri can help direct further diagnostic work — potentially reducing unnecessary investigations.
But Important Cautions Remain
Despite the excitement, experts urge prudence. While headline numbers are impressive, there are structural limitations and unanswered questions:
- The test missed a substantial fraction of cancers: overall “episode sensitivity” was 40.4% — meaning it detected only about four in ten cancers that emerged in the following year.
- Because sensitivity varies by cancer type and stage, the test is less reliable for some cancers — especially early-stage tumours or cancers that shed less cfDNA.
- A positive result does not equate to a definitive cancer diagnosis — it requires follow-up imaging or diagnostic confirmation. False positives, though rare, still occur.
- Also, a negative result does not guarantee absence of cancer, particularly for tumours not shedding enough DNA or not detectable by the test.
- Most importantly: there is currently no evidence that using Galleri — or any multi-cancer early detection (MCED) test — reduces cancer mortality. Experts emphasize that randomised trials with mortality as endpoint are needed before widespread adoption.
What Experts Say
Proponents of Galleri argue the test could “fundamentally change” cancer screening by extending it beyond a handful of cancers. Meanwhile, critics caution that the promise must be balanced against the risks of overdiagnosis, false reassurance from negative tests, and the uncertain impact on long-term outcomes such as survival.
As one recent review pointed out, while MCED tests are rapidly evolving, “screening interventions that are done on very broad swaths of the population — most of whom will not benefit — should be very well characterized,” including benefits, harms, costs, and downstream consequences.
References
- Exciting results from blood test for 50 cancers – BBC – (Accessed on Nov 27, 2025)
- Exciting’ New Cancer Blood Test Results Hide a Concerning Problem – Science Alert – (Accessed on Nov 27, 2025)
- Putting early cancer detection to the test – Nature – (Accessed on Nov 27, 2025)
- The Catch in Catching Cancer Early – The New Yorker – (Accessed on Nov 27, 2025)