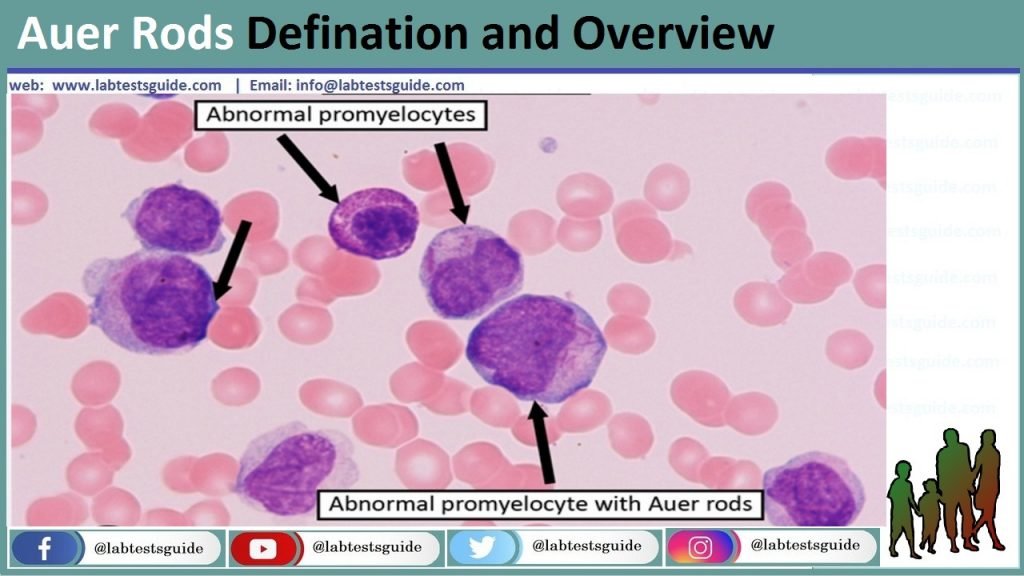
Auer rods are microscopic structures that are often seen in the cytoplasm of certain abnormal white blood cells, specifically myeloblasts, which are immature cells of the myeloid lineage. These rods are needle-like or rod-shaped inclusions and are typically stained red when a special staining technique called Wright’s stain is used.
What Are Auer Rods?
Auer rods are needle-like or rod-shaped structures that are found within the cytoplasm of certain abnormal white blood cells, specifically myeloblasts. These structures are a distinctive feature of a group of hematologic disorders known as acute myeloid leukemia (AML).
Auer Rods in Hematologic Disorders:
- Acute Myeloid Leukemia (AML): Auer rods are most commonly observed in AML, a type of blood cancer that affects myeloid cells in the bone marrow. In AML, immature myeloblasts accumulate in the bone marrow and bloodstream, and these cells often contain Auer rods. The presence of Auer rods is a diagnostic criterion for AML and can help distinguish it from other types of leukemia.
- Subtypes of AML: Auer rods can provide further information about the subtype and classification of AML. Different subtypes of AML may have distinct clinical characteristics and prognoses, and the presence or absence of Auer rods in myeloblasts can help classify AML into specific subtypes, such as acute promyelocytic leukemia (APL), which is associated with a particular genetic abnormality (t(15;17) translocation) and responds well to specific treatments.
- Other Myeloid Neoplasms: While AML is the most common hematologic disorder associated with Auer rods, these structures can also rarely be found in other myeloid neoplasms, such as myelodysplastic syndromes (MDS) and myeloproliferative neoplasms (MPN). In these conditions, the presence of Auer rods may be less frequent but can still provide diagnostic clues.
- Monitoring Disease Progression: Auer rods may be monitored over the course of treatment for hematologic disorders. Their presence or absence in myeloblasts can be used to assess the effectiveness of therapeutic interventions and to track disease progression or remission.
- Treatment Considerations: The presence of Auer rods can influence treatment decisions for AML. Some AML subtypes, such as APL, may require specific targeted therapies, such as all-trans retinoic acid (ATRA), which can induce differentiation of leukemic cells and lead to the disappearance of Auer rods.
Morphology and Structure:
- Shape and Size: Auer rods are typically elongated, needle-like, or rod-shaped structures. They can vary in size but are generally small and appear as fine, thread-like or slightly thicker structures within the cytoplasm of myeloblasts. The length of Auer rods can range from a few micrometers to several micrometers.
- Coloration: When subjected to special staining techniques, such as Wright’s stain or other Romanowsky-type stains, Auer rods often take on a characteristic reddish-purple or pink color. This staining pattern is crucial for their identification under a microscope.
- Composition: Auer rods are composed of aggregated granules and cellular material. These granules are derived from abnormal granulocytic or myelocytic precursor cells. The exact composition of Auer rods may vary, but they typically contain proteins, enzymes, and other cellular components that are part of the disrupted maturation process of myeloid cells.
- Intracellular Location: Auer rods are located within the cytoplasm of myeloblasts. They are not found in the nucleus of the cell but rather appear as inclusions within the cytoplasmic compartment.
- Variability: Auer rods can vary in appearance between different cases of AML and even within the same case. They may be more or less prominent, and their numbers can vary. Some AML subtypes, such as acute promyelocytic leukemia (APL), may have a higher prevalence of Auer rods and more distinctive-looking rods due to specific genetic abnormalities.
- Association with Malignancy: Auer rods are considered a hallmark of malignancy in myeloid cells. Their presence is indicative of a disruption in the normal maturation of white blood cells, particularly myeloblasts, and is strongly associated with hematologic malignancies like AML.
Causes of Auer Rods:
Here are some of the causes and factors contributing to the formation of Auer rods.
- Genetic Abnormalities: Auer rods are frequently observed in AML cases that have specific genetic abnormalities. The most well-known genetic alteration associated with Auer rods is the t(15;17) translocation, which is characteristic of acute promyelocytic leukemia (APL). This translocation results in the fusion of the PML and RARA genes and plays a central role in APL pathogenesis. The resulting abnormal protein product disrupts normal myeloid cell differentiation and contributes to Auer rod formation.
- Chromosomal Aberrations: Besides the t(15;17) translocation, other chromosomal abnormalities in AML, such as inv(16), t(8;21), and complex karyotypes, can also lead to the formation of Auer rods. These genetic alterations affect various genes involved in myeloid cell development, and their disruption can result in the accumulation of immature myeloblasts with Auer rods.
- Mutations in Regulatory Genes: Mutations in genes that regulate myeloid cell differentiation and maturation can contribute to Auer rod formation. For example, mutations in the CEBPA gene, which plays a role in granulocyte differentiation, are associated with AML and the presence of Auer rods.
- Disrupted Cellular Maturation: Auer rods are indicative of a block or arrest in the normal maturation process of myeloid cells. This disruption can be caused by various factors, including genetic mutations, chromosomal abnormalities, or alterations in signaling pathways that control cell differentiation.
- Hematologic Disorders: Auer rods are primarily found in hematologic disorders, particularly AML. However, they can also rarely occur in other myeloid neoplasms, such as myelodysplastic syndromes (MDS) and myeloproliferative neoplasms (MPN). In these cases, underlying genetic or molecular abnormalities contribute to the formation of Auer rods.
Symptoms of Auer Rods:
Common symptoms of AML include.
- Fatigue: Generalized fatigue and weakness are common symptoms of AML, often due to the decreased production of healthy red blood cells (anemia).
- Fever: Persistent or recurrent fever may occur as a result of infections, which can develop more easily when the number of normal white blood cells is reduced in AML.
- Frequent Infections: AML can lead to a weakened immune system, making individuals more susceptible to infections, such as bacterial, fungal, or viral infections.
- Bleeding and Bruising: AML can cause a decrease in platelet levels (thrombocytopenia), leading to easy bruising, nosebleeds, gum bleeding, or prolonged bleeding from minor cuts or injuries.
- Petechiae: Small, red or purple spots (petechiae) may appear on the skin or mucous membranes due to low platelet counts.
- Bone Pain: Some individuals with AML may experience bone pain or tenderness, which can result from the expansion of abnormal cells in the bone marrow.
- Swollen Lymph Nodes: Enlarged lymph nodes (lymphadenopathy) may be present, particularly in cases where the leukemia has spread to the lymph nodes.
- Splenomegaly: An enlarged spleen (splenomegaly) can occur in some cases of AML, leading to abdominal discomfort or fullness.
- Weight Loss: Unintended weight loss may be a symptom of AML, often due to a loss of appetite and the metabolic effects of the disease.
- Shortness of Breath: AML can lead to reduced oxygen-carrying capacity of the blood, resulting in shortness of breath.
Diagnosis of Auer Rods:
- Clinical Assessment: The diagnostic process often begins with a thorough medical history and physical examination. The healthcare provider will inquire about the patient’s symptoms, medical history, and any relevant risk factors.
- Complete Blood Count (CBC): A CBC is a blood test that provides important information about the number and types of blood cells in the bloodstream. In AML, abnormalities in the CBC may include low red blood cell counts (anemia), low platelet counts (thrombocytopenia), and abnormal white blood cell counts.
- Peripheral Blood Smear: A peripheral blood smear involves examining a thin layer of a patient’s blood under a microscope. This test can reveal the presence of abnormal white blood cells, including myeloblasts with Auer rods. The appearance of Auer rods in these cells is highly suggestive of AML.
- Bone Marrow Aspiration and Biopsy: To confirm the diagnosis of AML and to classify it into specific subtypes, a bone marrow aspiration and biopsy are typically performed. During a bone marrow aspiration, a small sample of bone marrow is withdrawn from the hipbone or another large bone using a needle. A bone marrow biopsy involves the removal of a core of bone and marrow tissue. Both samples are then examined under a microscope to assess the presence of Auer rods and the proportion of abnormal cells.
- Cytogenetic and Molecular Studies: Specialized tests, such as cytogenetic analysis and molecular genetic testing, are conducted to identify specific genetic abnormalities and mutations associated with AML. These tests can help determine the subtype of AML and guide treatment decisions. For example, the t(15;17) translocation is associated with acute promyelocytic leukemia (APL), a subtype of AML, and requires distinct treatment approaches.
- Imaging Studies: Imaging studies, such as X-rays, CT scans, or ultrasound, may be performed to evaluate the extent of disease involvement and assess any organ enlargement, such as an enlarged spleen or lymph nodes.
- Lumbar Puncture: In some cases, a lumbar puncture (spinal tap) may be recommended to assess whether the leukemia has spread to the cerebrospinal fluid and the central nervous system.
- Flow Cytometry: Flow cytometry is a technique that uses fluorescent antibodies to identify and classify specific types of blood cells, including abnormal ones. It can help confirm the diagnosis and determine the immunophenotype of the leukemia cells.
- Histopathology and Immunohistochemistry: If a tissue biopsy is obtained from an extramedullary site (outside the bone marrow), histopathology and immunohistochemistry may be used to examine the tissue and determine if it contains leukemia cells.
Prevents of Auer Rods:
Here are some general strategies for preventing AML.
- Healthy Lifestyle: Maintaining a healthy lifestyle can contribute to overall well-being and reduce the risk of various health conditions, including some forms of leukemia. This includes a balanced diet, regular exercise, adequate sleep, and stress management.
- Avoiding Smoking: Smoking tobacco is a known risk factor for AML. Avoiding or quitting smoking can significantly reduce the risk of developing AML and other smoking-related cancers.
- Chemical Exposure: Minimizing exposure to certain chemicals and toxins that are known or suspected carcinogens may reduce the risk of AML. Occupational exposures to chemicals like benzene and formaldehyde have been linked to AML. Following safety guidelines and using protective equipment in workplaces with potential chemical hazards can help mitigate risks.
- Radiation Exposure: High levels of ionizing radiation exposure, such as from atomic bomb detonations or certain medical treatments, are associated with an increased risk of AML. Efforts to limit unnecessary exposure to radiation, especially in medical settings, can help reduce this risk.
- Genetic Counseling: Some individuals may have a family history of AML or other hematologic disorders. Genetic counseling can help individuals understand their familial risk factors and make informed decisions about their health.
- Viral Infections: Certain viral infections, such as human T-cell lymphotropic virus type 1 (HTLV-1), have been associated with an increased risk of AML. Reducing the risk of these infections through safe practices and vaccination, when available, may help.
- Chemotherapy-Related AML: Some cases of AML are secondary to chemotherapy or radiation therapy for other cancers. In these cases, healthcare providers carefully consider the treatment plan and potential risks when using these therapies.
Treatment and Management:
- Chemotherapy: Chemotherapy is the primary treatment for AML. It involves the use of powerful medications to kill leukemia cells or prevent their further growth and division. Different chemotherapy regimens may be used, and the choice depends on the patient’s age, general health, and the specific characteristics of the leukemia cells.
- Induction Therapy: The initial phase of treatment, called induction therapy, aims to achieve remission. Patients typically receive a combination of chemotherapy drugs. Common regimens include 7+3 (cytarabine and an anthracycline, such as daunorubicin or idarubicin).
- Consolidation Therapy: After achieving remission, consolidation therapy is administered to eliminate any remaining leukemia cells and reduce the risk of relapse. This may involve additional cycles of chemotherapy.
- Targeted Therapy: In some cases, targeted therapies are used to specifically target and inhibit proteins or genetic mutations associated with AML. For example, tyrosine kinase inhibitors like imatinib may be used for patients with specific genetic mutations, such as the FLT3-ITD mutation.
- Stem Cell Transplantation: Stem cell transplantation, also known as a bone marrow transplant or hematopoietic stem cell transplant, may be considered for certain AML patients. This procedure involves replacing the patient’s diseased bone marrow with healthy stem cells from a matched donor (allogeneic transplant) or from the patient’s own stem cells (autologous transplant). Stem cell transplantation is typically reserved for individuals who have a high risk of relapse or those who have relapsed after initial treatment.
- Supportive Care: AML treatment often leads to side effects, such as low blood cell counts, infections, and fatigue. Supportive care measures, including blood transfusions, antibiotics, and medications to stimulate the production of blood cells, help manage these side effects and improve the patient’s quality of life during treatment.
- Clinical Trials: Participation in clinical trials may be an option for some AML patients, particularly those with relapsed or refractory disease. Clinical trials offer access to experimental treatments and therapies that may have the potential to improve outcomes.
- Monitoring: After treatment, regular follow-up visits and monitoring are crucial to assess the patient’s response to treatment, detect any signs of relapse, and manage potential long-term side effects of treatment.
- Palliative Care: In cases where AML is advanced and curative treatment options are limited, palliative care can provide relief from symptoms, improve quality of life, and offer emotional support to both patients and their families.
Research and Advancements:
Some of the recent research areas and advancements in AML include.
- Genomic Profiling: Advances in genomic sequencing technologies have allowed for more comprehensive profiling of AML patients’ genetic and molecular characteristics. This has led to the discovery of novel mutations and genetic abnormalities that can help guide treatment decisions and predict outcomes. Genomic profiling has also identified potential therapeutic targets, leading to the development of targeted therapies for specific AML subtypes.
- Targeted Therapies: Targeted therapies have emerged as promising treatment options for AML. Drugs like venetoclax, gilteritinib, and ivosidenib have been approved for use in specific AML subtypes or genetic mutations. These therapies selectively target the molecular abnormalities driving AML cells, often resulting in improved response rates and reduced side effects compared to traditional chemotherapy.
- Immunotherapy: Immunotherapeutic approaches, including immune checkpoint inhibitors and chimeric antigen receptor T-cell (CAR-T) therapy, are being investigated in clinical trials for AML. These treatments aim to harness the body’s immune system to recognize and attack leukemia cells.
- Minimal Residual Disease (MRD) Monitoring: MRD refers to the small number of cancer cells that may remain in a patient’s body after treatment and are undetectable by standard tests. Improved techniques for MRD monitoring allow for more sensitive detection of residual leukemia cells, enabling more accurate assessment of treatment response and risk of relapse.
- Stem Cell Transplantation: Research is ongoing to optimize the use of stem cell transplantation in AML, including identifying the most suitable transplant donors and refining transplant techniques to reduce complications and improve outcomes.
- Novel Drug Combinations: Researchers are exploring combinations of targeted therapies, chemotherapy, and other agents to improve treatment outcomes in AML. Combinations that enhance the efficacy of standard therapies while minimizing side effects are of particular interest.
- Personalized Medicine: A growing focus in AML research is the development of personalized treatment strategies based on a patient’s unique genetic and molecular profile. Tailored treatment plans aim to optimize therapeutic benefit while minimizing the risk of treatment-related complications.
- Supportive Care: Research continues to improve supportive care measures, such as infection prevention, management of treatment-related side effects, and psychosocial support, to enhance the overall well-being of AML patients during and after treatment.
- Clinical Trials: Participation in clinical trials is essential for testing novel treatments and therapies in AML. Clinical trials provide opportunities for patients to access innovative treatments and contribute to advancing the field.
- Biomarker Discovery: Ongoing research efforts aim to identify new biomarkers that can aid in early diagnosis, risk stratification, and treatment selection for AML patients.
FAQs:
What are Auer rods?
Auer rods are needle-like or rod-shaped structures found in the cytoplasm of certain abnormal white blood cells, particularly myeloblasts. They are indicative of a group of hematologic disorders, primarily acute myeloid leukemia (AML).
What is acute myeloid leukemia (AML)?
AML is a type of blood cancer that begins in the bone marrow and affects myeloid cells, leading to the rapid growth of abnormal white blood cells (myeloblasts). Auer rods are a characteristic feature of AML.
How are Auer rods detected and diagnosed?
Auer rods are detected through microscopic examination of blood or bone marrow samples, usually stained with special techniques like Wright’s stain. Their presence confirms the diagnosis of AML and helps classify the disease into specific subtypes.
What are the common symptoms of AML?
Common symptoms of AML include fatigue, fever, frequent infections, bleeding or bruising easily, bone pain, swollen lymph nodes, weight loss, and shortness of breath. These symptoms result from disruptions in normal blood cell production.
What is the treatment for AML?
Treatment for AML typically involves chemotherapy, including induction and consolidation therapy. Other options may include targeted therapy, stem cell transplantation, and supportive care to manage side effects and complications.
Are there preventive measures for Auer rods or AML?
Auer rods themselves are a diagnostic feature, not a condition. Preventive measures for AML include a healthy lifestyle, avoiding smoking, minimizing exposure to chemicals, and being cautious with radiation exposure.
What recent advancements have been made in AML treatment?
Recent advancements in AML treatment include targeted therapies, immunotherapies, personalized medicine, improved stem cell transplantation techniques, and novel drug combinations. Genomic profiling has also led to a better understanding of the disease and potential therapeutic targets.
How can I stay informed about AML research and treatments?
Staying informed about AML involves regularly consulting healthcare professionals, following reputable medical websites and journals, participating in patient support groups, and considering enrollment in clinical trials to access cutting-edge treatments and contribute to research.
Conclusion:
In conclusion, Auer rods are distinctive needle-like or rod-shaped structures found within the cytoplasm of abnormal myeloblasts and are a defining feature of acute myeloid leukemia (AML). Their identification plays a crucial role in diagnosing AML, determining its subtype, and guiding treatment decisions. Recent advancements in AML research have led to more targeted therapies, personalized treatment approaches, and improved outcomes for patients. While AML remains a serious hematologic disorder, ongoing research continues to shape our understanding of the disease, offering hope for better treatments and outcomes in the future.