Real-time PCR (quantitative PCR, qPCR) is now a well-established method for the detection, quantification and typing of different microbial agents in the areas of clinical and veterinary diagnostics and food safety. Although the concept of PCR is relatively simple, there are specific issues in qPCR that developers and users of this technology must be aware of. These include the use of correct terminology and definitions, understanding of the principle of PCR, difficulties with interpretation and presentation of data, limitations of qPCR in different areas of microbial diagnosis, and parameters important for the description of qPCR performance. It is not our intention in this review to describe each and every aspect of qPCR design, optimization and validation; however, we hope this basic guide will help guide qPCR beginners and users in the use of this powerful technique.
- It is a technique used to monitor the progress of a PCR reaction in real time.
- At the same time, a relatively small amount of PCR product (DNA, cDNA or RNA) can be quantified.
- It is based on the detection of the fluorescence produced by a reporter molecule that increases as the reaction progresses.
- It is also known as quantitative polymerase chain reaction (qPCR), which is a molecular biology laboratory technique based on the polymerase chain reaction (PCR).
- qPCR is a powerful technique that allows exponential amplification of DNA sequences.
- A PCR reaction needs a pair of primers that are complementary to the sequence of interest. The primers are extended by DNA polymerase.
- The copies produced after extension, the so-called amplicons, are re-amplified with the same primers, leading to an exponential amplification of the DNA molecules.
- However, after amplification, gel electrophoresis is used to analyze the amplified PCR products and this makes conventional PCR time consuming; as the reaction must end before proceeding with the post-PCR analysis. Real-time PCR solves this problem.
- The term “real-time” denotes that you can monitor the progress of amplification when the process is in progress, in contrast to the conventional PCR method where analysis is possible only after the process is complete.
PCR terminology
| Polymerase chain reaction | PCR |
| Reverse transcription-polymerase chain reaction | RT-PCR |
| Real-time polymerase chain reaction | qPCR |
| RT-PCR / qPCR combined technique | qRT-PCR |

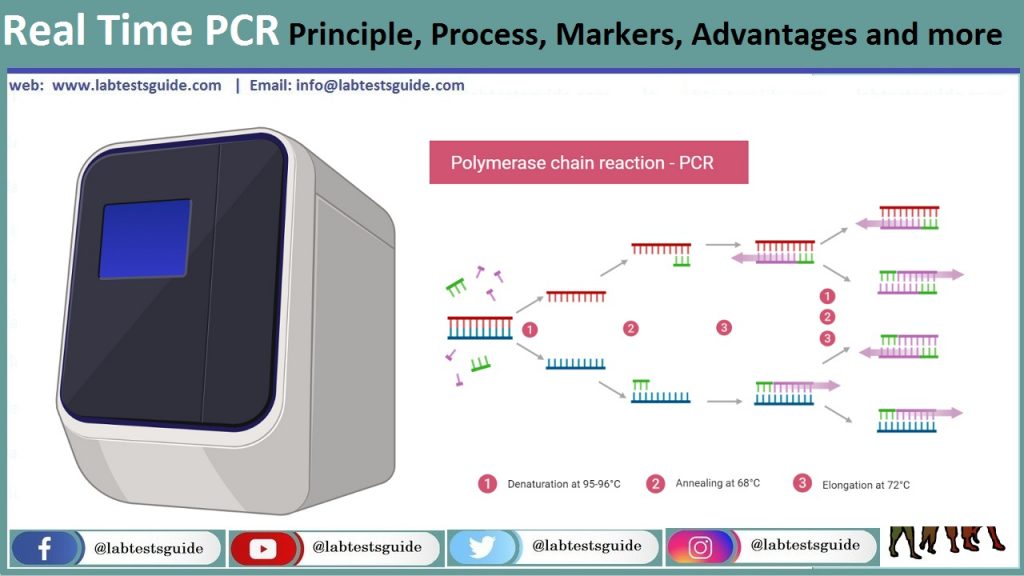
Real Time PCR Principle
Este mismo principio de amplificación de PCR se emplea en PCR en tiempo real. Pero en lugar de mirar las bandas en un gel al final de la reacción, el proceso se monitorea en “tiempo real”. La reacción se coloca en una máquina de PCR en tiempo real que observa cómo ocurre la reacción con una cámara o detector.
Aunque se utilizan muchas técnicas diferentes para controlar el progreso de una reacción de PCR, todas tienen una cosa en común. Todos ellos vinculan la amplificación del ADN con la generación de fluorescencia que puede detectarse simplemente con una cámara durante cada ciclo de PCR. Por lo tanto, a medida que aumenta el número de copias de genes durante la reacción, también lo hace la fluorescencia, lo que indica el progreso de la reacción.
Steps of Real Time PCR (Protocol)
The working procedure can be divided into two steps:
A. Amplification
Denaturation
High-temperature incubation is used to “melt” the double-stranded DNA into single-stranded and loosen the secondary structure in the single-stranded DNA. Typically the highest temperature that DNA polymerase can withstand is used (typically 95 ° C). The denaturation time can be increased if the content of the GC template is high.
Annealing:
During hybridization, complementary sequences have the opportunity to hybridize, so an appropriate temperature is used which is based on the calculated melting temperature (Tm) of the primers (5 ° C below the Tm of the primer).
Extension:
At 70-72 ° C, DNA polymerase activity is optimal and primer extension occurs at rates of up to 100 bases per second. When an amplicon in real-time PCR is small, this step is often combined with the annealing step using 60 ° C as the temperature.
B. Detection
- Detection is based on fluorescence technology.
- The sample is first kept in the appropriate well and thermally cycled as in normal PCR.
- The machine, however, in real-time PCR is subjected to a source of tungsten or halogen which causes the fluorescent marker to add to the sample and the signal is amplified with the amplification of the copy number of the DNA sample.
- The emitted signal is detected by a detector and sent to the computer after conversion to a digital signal that is displayed on the screen.
- The signal can be detected when the threshold level rises (lowest detection level of the detector)
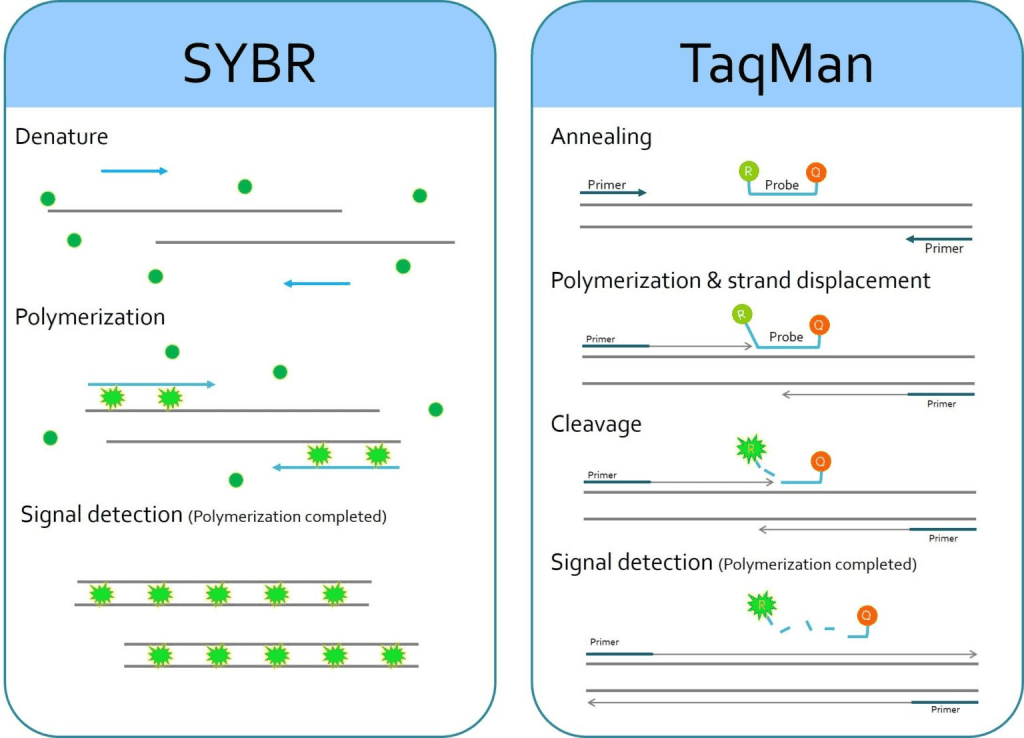
Fluorescence Markers used in Real Time PCR

There are many different markers used in Real Time PCR but the most common of them include:
- Taqman probe.
- SYBR Green.
Taqman Probe
- It is a hydrolysis probe that carries an indicator dye, often fluorescein (FAM) at its 5 ‘end and a tetramethylrhodamine inhibitor (TAMRA), attached to the 3’ end of the oligonucleotide.
- Under normal conditions, the probe remains coiled on itself carrying the fluorescence dye close to the quencher, which inhibits or turns off the fluorescent signal of the dye.
- The Taq polymerase oligonucleotide has a region homologous to the target gene and therefore, when the target sequence is present in the mixture, it binds to the DNA of the sample.
- As the taqpolymerase begins to form a new strand of DNA in the extension stage, it causes degradation of the probe by the nuclease activity of the 5 ‘end and the fluorescein is separated from the quencher as a result of which the signal of fluorescence.
- As this procedure continues, in each cycle the number of signal molecules increases, causing an increase in fluorescence that is positively related to the amplification of the target.
SYBR Green
- This is a dye that emits a prominent fluorescent signal when it binds to the minor groove of DNA, in a nonspecific way.
- Other fluorescent dyes such as ethidium bromide or acridine orange can also be used, but SYBR Green is best used because of its higher signal intensity.
- SYBR Green is more preferred than the Taqman probe as it can provide information on each amplification cycle as well as melting temperature that is not obtained from the Taqman probe.
- However, its downside is the lack of specificity compared to the Taqman Probe.
Advantages:
It has many advantages over normal PCR:
- Take a look at the reaction to help decide which reactions have worked well and which have failed.
- The efficiency of the reaction can be calculated precisely.
- It is not necessary to run the PCR product on a gel after the reaction, as melting curve analysis serves that purpose.
- Real-time PCR data can be used for truly quantitative analysis of gene expression. By comparison, old-fashioned PCR was only semi-quantitative at best.
- PCR faster than normal.
- Less complexity in quantifying sample.etc.
Thus, unlike ordinary preparative PCR, real-time PCR makes it possible to automatically determine the success of a multiplex PCR reaction after a few cycles, without a separate analysis of each reaction, and avoids the problem of “false negatives. ”.
Applications
- Gene expression analysis
- Cancer research
- Drug research
- Disease diagnosis and management
- Viral quantification
- Food testing
- GMO food
- Animal and plant breeding
- Gene copy number
References
- https://www.mun.ca/biology/scarr/Principle_of_RT-PCR.html
- http://www.primerdesign.co.uk/assets/files/beginners_guide_to_real_time_pcr.pdf
- https://link.springer.com/chapter/10.1007%2F978-90-481-3132-7_3
- https://www.slideshare.net/pratyayseth/real-time-pcr-34159486
- https://www.thermofisher.com/np/en/home/life-science/pcr/real-time-pcr/real-time-pcr-learning-center/real-time-pcr-basics/essentials-real-time-pcr.html
- https://www.sciencedirect.com/topics/neuroscience/real-time-polymerase-chain-reaction
- https://microbenotes.com/real-time-pcr-principle-process-markers-advantages-applications/