Enzymatic assays are laboratory techniques used to measure the activity of enzymes or the concentration of specific substrates, products, or coenzymes in biological samples. Enzymes are essential proteins that catalyze biochemical reactions in living organisms, and their activity can provide important insights into various biological processes, disease mechanisms, and drug development. There are several types of enzymatic assays, each designed for specific purposes.

Definition of Enzymatic Assays:
Enzymatic assays refer to a set of laboratory techniques and procedures used to quantitatively or qualitatively measure the activity of enzymes or determine the concentration of specific substrates, products, or coenzymes involved in enzymatic reactions. These assays are crucial for studying enzyme function, characterizing their properties, and understanding various biological, biochemical, and medical processes.
Importance of Enzymatic Assays:
- Biological Research: Enzymatic assays are fundamental tools in biological research, allowing scientists to investigate and elucidate the roles of enzymes in various cellular processes. They provide insights into enzyme kinetics, mechanisms of action, and regulatory pathways.
- Drug Discovery and Development: Enzymatic assays play a pivotal role in pharmaceutical research. They are used to screen potential drug candidates by testing their effects on specific enzymes. Understanding enzyme inhibition or activation is crucial for designing drugs to treat diseases.
- Clinical Diagnostics: Enzymatic assays are used in clinical laboratories for diagnosing various medical conditions. They can detect specific biomarkers or enzymes associated with diseases, aiding in early disease detection and monitoring patient health.
- Biotechnology: Enzymatic assays are used in biotechnology to optimize and control bioprocesses. They are crucial for measuring enzyme activities in industrial applications like biofuel production, food processing, and biopharmaceutical manufacturing.
- Quality Control: In industries such as food and beverage, enzymatic assays are employed to monitor and ensure product quality. For example, they can be used to assess the enzymatic activity in food products like dairy and fruit juices.
- Environmental Monitoring: Enzymatic assays can be adapted to assess environmental conditions, such as measuring pollution levels in water or soil. Enzymes like catalase and peroxidase can be used as indicators of environmental health.
- Research in Genetics and Molecular Biology: Enzymatic assays are integral to genetic research, where they help study DNA and RNA polymerases, restriction enzymes, and DNA-modifying enzymes. They are also used in techniques like PCR (Polymerase Chain Reaction).
- Enzyme Engineering: Enzymatic assays are essential in the field of enzyme engineering, where researchers modify enzymes for specific applications. Assays help determine the efficiency and specificity of engineered enzymes.
- Biomedical Research: Enzymatic assays are used in studying cellular pathways, biomarker discovery, and understanding disease mechanisms. They contribute to advancements in personalized medicine and targeted therapies.
- Teaching and Education: Enzymatic assays are valuable for teaching biochemistry and molecular biology concepts in academic settings, allowing students to gain hands-on experience with fundamental laboratory techniques.
Types of Enzymatic Assays:
- Spectrophotometric Assays: These assays measure changes in absorbance of light at specific wavelengths to track enzyme activity. They are versatile and widely used due to their ability to monitor a wide range of enzymes.
- Fluorometric Assays: Fluorometric assays detect changes in fluorescence emission during an enzymatic reaction, offering high sensitivity and suitability for low-activity enzymes.
- Radioactive Assays: Radioactive isotopes are used to label substrates or products, allowing for highly sensitive detection and quantification of enzyme activity. Safety precautions are essential.
- Colorimetric Assays: These assays rely on the formation of a colored product during the enzymatic reaction, and the color intensity is proportional to enzyme activity. They are straightforward and commonly used.
- Chromatographic Assays: Techniques like HPLC and GC separate and quantify substrates, products, or coenzymes involved in enzymatic reactions, providing precise measurements.
- ELISA (Enzyme-Linked Immunosorbent Assay): ELISA uses enzyme-conjugated antibodies to detect specific antigens or antibodies in biological samples, making it valuable in clinical diagnostics and immunology.
- Enzyme Activity Assays: These assays directly measure the rate of product formation or substrate consumption, providing a straightforward assessment of enzyme activity.
- Enzyme Kinetics Assays: Enzyme kinetics studies the rate of enzyme-catalyzed reactions as a function of various factors, helping determine kinetic parameters and reaction mechanisms.
- Molecular Biology Techniques: Enzymatic assays in molecular biology analyze DNA and RNA processing enzymes, such as DNA polymerases and nucleases, aiding in DNA replication and gene expression studies.
- Immunoassays: Enzyme-based immunoassays like ELISAs and RIAs detect specific antigens or antibodies, playing a crucial role in diagnosing diseases and monitoring immune responses.
Applications of Enzymatic Assays:
- Drug Discovery and Development: Enzymatic assays are used to screen and evaluate potential drug candidates for their effects on specific enzymes. This helps identify compounds that can be used to modulate enzyme activity, which is essential for designing new drugs.
- Clinical Diagnostics: Enzymatic assays are employed in clinical laboratories to diagnose and monitor various diseases and medical conditions. They can detect specific biomarkers or enzymes associated with conditions like heart disease, liver dysfunction, and diabetes.
- Biotechnology and Bioengineering: Enzymatic assays are crucial for optimizing and controlling bioprocesses in biotechnology applications. They are used to monitor enzyme activity in industrial processes such as biofuel production, food processing, and pharmaceutical manufacturing.
- Quality Control in Food and Beverage Industry: Enzymatic assays are used to assess the quality of food and beverage products. For example, they can determine the enzyme activity in dairy products, fruit juices, and brewing processes.
- Environmental Monitoring: Enzymatic assays can be adapted for environmental purposes, such as measuring pollution levels in water, soil, and air. Enzymes like catalase and peroxidase can serve as indicators of environmental health.
- Genetics and Molecular Biology Research: Enzymatic assays are essential in genetic research, allowing scientists to study DNA and RNA polymerases, restriction enzymes, and DNA-modifying enzymes. They are also used in techniques like PCR (Polymerase Chain Reaction).
- Biomedical Research: Enzymatic assays aid in studying cellular pathways, discovering biomarkers, and understanding disease mechanisms. They contribute to advances in personalized medicine and targeted therapies.
- Enzyme Engineering: Researchers use enzymatic assays to assess the efficiency and specificity of engineered enzymes. This is critical for designing enzymes with improved properties for various applications.
- Teaching and Education: Enzymatic assays are valuable for teaching biochemistry and molecular biology concepts in educational settings. Students can gain hands-on experience with fundamental laboratory techniques.
- Pharmacokinetics and Pharmacodynamics: Enzymatic assays are used in pharmacokinetic and pharmacodynamic studies to understand how drugs are metabolized and how they affect enzyme activity in the body. This information is vital for dosing and treatment strategies.
- Nutritional Analysis: Enzymatic assays are employed in nutritional science to determine the nutrient content of food products and assess their bioavailability.
- Agricultural and Crop Sciences: Enzymatic assays are used in agriculture to study enzymes involved in plant growth, development, and stress responses. They are also employed in crop protection and pest control strategies.
- Enzyme-based Biosensors: Enzymatic assays are integrated into biosensors for real-time monitoring of specific analytes in fields like environmental monitoring, healthcare, and food safety.
Principles of Enzymatic Assays:
The principles of enzymatic assays are fundamental concepts that underlie the design and execution of experiments to measure enzyme activity or determine the concentration of specific substrates, products, or coenzymes in biological samples. Here are the key principles of enzymatic assays:
- Specificity: Enzymatic assays are designed to measure a specific enzyme’s activity or a particular substrate/product of interest. The assay components, including substrates, cofactors, and detection reagents, should be carefully chosen to target the enzyme or molecule of interest selectively.
- Enzyme-Substrate Interaction: Enzymes catalyze reactions by binding to their substrates, forming enzyme-substrate complexes. Enzymatic assays rely on this interaction, and the rate of product formation or substrate consumption is proportional to the enzyme’s activity.
- Reaction Kinetics: Enzymatic reactions follow kinetic principles, including the Michaelis-Menten equation. Understanding the kinetics helps determine the initial reaction rates, substrate saturation, and enzyme affinity for substrates.
- Linearity: Enzymatic assays should operate under conditions where the reaction rates are linear with respect to time and substrate or enzyme concentration. This allows for accurate measurements and calculations.
- Standardization: To ensure consistency and reliability, enzymatic assays often require standardization using known concentrations of substrates, products, or reference enzymes with known activity levels. Standard curves can be used for quantification.
- Controls: Including appropriate controls is essential to validate the assay. Negative controls without the enzyme or substrates help identify background noise, while positive controls with known enzyme activity validate the assay’s performance.
- Detection Methods: Enzymatic assays employ various detection methods, such as spectrophotometry, fluorometry, or radioactive counting, depending on the nature of the enzyme and the reaction. Detection relies on changes in absorbance, fluorescence, or radioactivity.
- Reaction Conditions: Enzymatic assays require optimal reaction conditions, including temperature, pH, and ionic strength, to mimic physiological or experimental conditions accurately. These conditions can significantly impact enzyme activity.
- Data Analysis: Accurate data analysis is crucial. Enzymatic assays involve calculating reaction rates, substrate concentrations, and enzyme activity based on experimental measurements. Kinetic analysis often involves plotting data and fitting curves to mathematical models.
- Inhibition and Activation: Enzymatic assays can be used to study enzyme inhibition or activation. Inhibitors can be tested to determine their effects on enzyme activity, aiding drug discovery efforts.
- Stoichiometry: Understanding the stoichiometry of the reaction (the ratio of reactants to products) is essential for precise quantification of enzyme activity or substrate concentration.
- Safety: Enzymatic assays that involve radioactive or hazardous materials must adhere to safety protocols and regulations. Proper disposal and handling of materials are crucial.
- Calibration and Validation: Enzymatic assays should be calibrated and validated regularly to ensure their accuracy and reliability. This includes using known standards and controls to confirm assay performance.
- Optimization: Experimenters often optimize assay conditions, such as enzyme concentration, reaction time, or substrate concentration, to maximize sensitivity and precision.
- Reproducibility: Ensuring the reproducibility of enzymatic assays is essential. Proper documentation of protocols and quality control measures helps achieve consistent results across experiments.
Common Enzymatic Assay Techniques:
Several common enzymatic assay techniques are used in laboratory research, clinical diagnostics, and various industries to measure enzyme activity or the concentration of specific substrates, products, or coenzymes. Here are some of the most commonly used enzymatic assay techniques:
- Spectrophotometric Assays:
- UV-Visible Spectrophotometry: Measures changes in light absorbance at specific wavelengths during the enzymatic reaction, allowing quantification of substrate consumption or product formation.
- Colorimetric Assays: Utilizes the formation of colored products during the reaction, with the color intensity proportional to enzyme activity. Common examples include the Bradford assay for protein quantification and the alkaline phosphatase assay.
- Fluorometric Assays:
- Fluorescence Spectroscopy: Monitors changes in fluorescence emission as a result of enzymatic reactions, providing high sensitivity and specificity. Commonly used for studying nucleic acid amplification and enzyme kinetics.
- Radioactive Assays:
- Radioactivity Measurement: Involves labeling substrates or products with radioactive isotopes (e.g., ³H, ¹⁴C) and quantifying radioactivity levels using scintillation counters. Highly sensitive but requires stringent safety measures.
- Chromatographic Assays:
- High-Performance Liquid Chromatography (HPLC): Separates and quantifies substrates, products, or coenzymes based on their differential migration through a chromatographic column. Often used in pharmaceutical and biochemical research.
- Gas Chromatography (GC): Analyzes volatile compounds, including those involved in enzymatic reactions, by separating them based on their vapor pressure and chemical properties.
- ELISA (Enzyme-Linked Immunosorbent Assay):
- Direct ELISA: Detects and quantifies antigens or antibodies by employing enzyme-conjugated antibodies.
- Indirect ELISA: Utilizes a secondary antibody labeled with an enzyme for enhanced sensitivity. Often used in clinical diagnostics and immunological research.
- Enzyme Activity Assays:
- Initial Rate Assays: Measure the rate of product formation or substrate consumption during the early stages of the enzymatic reaction.
- Endpoint Assays: Determine the total product or substrate concentration at the end of the reaction.
- Enzyme Kinetics Assays:
- Michaelis-Menten Kinetics: Studies the relationship between substrate concentration and reaction rate, providing insights into enzyme efficiency and affinity.
- Lineweaver-Burk Plot: Graphical representation of enzyme kinetics data used to determine kinetic parameters like Km and Vmax.
- Molecular Biology Techniques:
- Polymerase Chain Reaction (PCR): Utilizes DNA polymerases to amplify specific DNA sequences. Enzymatic assays can be incorporated for quantification and analysis of PCR products.
- Nuclease Assays: Measure the activity of enzymes like restriction endonucleases and DNA/RNA nucleases, often used in molecular cloning and DNA manipulation.
- Immunoassays:
- Radioimmunoassay (RIA): Combines antibodies labeled with radioactive isotopes to detect and quantify antigens or antibodies.
- Enzyme Immunoassay (EIA): Utilizes enzyme-labeled antibodies for immunodetection, including ELISA mentioned earlier.
- Biochemical Assays:
- ATPase Assays: Measure the activity of enzymes that hydrolyze ATP (adenosine triphosphate).
- Protease Assays: Quantify the activity of proteolytic enzymes like proteases and peptidases.
Specialized Enzymatic Assay Methods:
Specialized enzymatic assay methods are tailored techniques designed to address specific research, diagnostic, or industrial needs. These methods often target particular enzymes or biochemical processes. Here are some examples of specialized enzymatic assay methods:
- Luciferase Assay:
- Bioluminescence Assay: Utilizes luciferase enzymes to produce light as a result of specific enzymatic reactions. Commonly used in reporter gene assays and ATP measurement assays.
- ATPase Assays:
- ATPase Activity Assay: Measures the hydrolysis of ATP by enzymes like ATPases. Useful in studying cellular energy metabolism and ion transport processes.
- Kinase Assays:
- Kinase Activity Assay: Measures the activity of protein kinases, which phosphorylate target proteins. Often used in drug development and signaling pathway studies.
- Phosphatase Assays:
- Phosphatase Activity Assay: Quantifies the activity of phosphatase enzymes, which remove phosphate groups from molecules. Important in cell signaling research.
- Glycosidase Assays:
- Glycosidase Activity Assay: Measures the activity of glycosidase enzymes involved in carbohydrate metabolism and glycan processing.
- Lipase Assays:
- Lipase Activity Assay: Quantifies the activity of lipase enzymes that hydrolyze lipids. Used in lipid metabolism research and quality control of food products.
- Amidase Assays:
- Amidase Activity Assay: Measures the activity of amidase enzymes that catalyze the hydrolysis of amide bonds in molecules.
- Protease Assays:
- Protease Activity Assay: Quantifies the activity of proteolytic enzymes (proteases) involved in protein degradation and processing.
- Polymerase Assays:
- Polymerase Activity Assay: Measures the activity of DNA or RNA polymerases involved in DNA replication, transcription, or reverse transcription. Important in molecular biology research and drug discovery.
- β-Galactosidase Assay:
- β-Galactosidase Activity Assay: Detects and quantifies the activity of β-galactosidase enzyme, commonly used in molecular biology reporter assays.
- Transaminase Assays:
- Transaminase Activity Assay: Measures the activity of transaminase enzymes, often used in clinical diagnostics and studying amino acid metabolism.
- Hyaluronidase Assay:
- Hyaluronidase Activity Assay: Quantifies the activity of hyaluronidase enzymes involved in the breakdown of hyaluronic acid.
- Glucose Oxidase Assay:
- Glucose Oxidase Activity Assay: Measures the activity of glucose oxidase enzymes, frequently used in glucose detection and biosensors.
- Alkaline Phosphatase Assay:
- Alkaline Phosphatase Activity Assay: Quantifies the activity of alkaline phosphatase, often used in molecular biology and clinical diagnostics.
- Coagulation Assays:
- Prothrombin Time (PT) and Activated Partial Thromboplastin Time (aPTT) Assays: Assess the coagulation cascade’s activity and clotting times, used in clinical hematology and diagnostics.
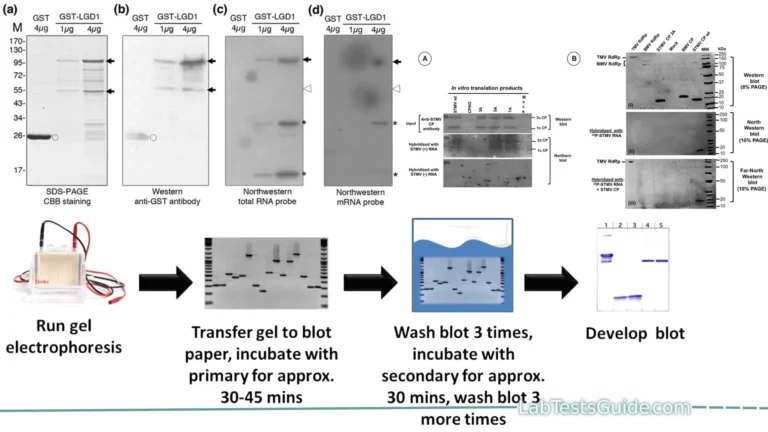
Experimental Techniques and Procedures:
Experimental techniques and procedures in enzymatic assays involve a series of steps and protocols designed to accurately measure enzyme activity or quantify specific substrates, products, or coenzymes. Below is an overview of common experimental techniques and procedures involved in enzymatic assays:
- Sample Preparation:
- Sample Collection: Obtain biological or chemical samples containing the enzyme of interest, substrates, and any necessary cofactors or activators.
- Sample Homogenization: If working with biological samples, homogenize or disrupt cells or tissues to ensure uniform distribution of enzymes and substrates.
- Sample Clarification: Centrifuge or filter samples to remove debris and particulate matter that may interfere with the assay.
- Reagent Preparation:
- Enzyme Solution: Prepare an enzyme solution of known concentration or activity, if not already commercially available.
- Substrate Solutions: Prepare substrate solutions at various concentrations if assessing enzyme kinetics.
- Control Solutions: Prepare positive and negative control solutions to validate assay performance.
- Buffer Solutions: Prepare buffers with the appropriate pH and ionic strength to maintain optimal enzyme activity.
- Assay Setup:
- Experimental Design: Plan the experiment, including the assay type (e.g., spectrophotometric, fluorometric, colorimetric), reaction conditions, and sample volumes.
- Reaction Vessel: Select appropriate containers, cuvettes, or microplates for the assay.
- Temperature Control: Maintain a constant temperature using water baths, heating blocks, or incubators, as temperature can significantly affect enzyme activity.
- Enzyme Reaction:
- Sample Addition: Add the enzyme solution, substrates, and any cofactors or activators to the reaction vessel.
- Start Timing: Initiate the reaction, start the timer, and record the time (t=0) when the reagents are mixed.
- Data Collection:
- Spectrophotometry: Measure absorbance or fluorescence at specific wavelengths at regular intervals to monitor changes over time.
- Sampling: At predefined time intervals, withdraw samples from the reaction vessel for analysis, or perform continuous monitoring.
- Standard Curve: If applicable, construct a standard curve using known concentrations of substrates or products to quantitate assay results.
- Data Analysis:
- Calculations: Use the collected data to calculate reaction rates, enzyme activity, substrate concentrations, or other relevant parameters.
- Graphical Analysis: Plot data on graphs, such as Michaelis-Menten plots or Lineweaver-Burk plots for kinetic analysis.
- Statistical Analysis: Perform statistical tests if comparing multiple conditions or replicates to assess significance.
- Quality Control:
- Controls: Include positive and negative controls to validate assay performance.
- Replicates: Perform assays in duplicate or triplicate to assess reproducibility.
- Calibration: Calibrate instruments, if applicable, to ensure accurate measurements.
- Safety Precautions:
- Safety Equipment: Wear appropriate personal protective equipment, such as lab coats, gloves, and safety goggles.
- Chemical Safety: Follow safety protocols for handling chemicals, particularly if radioactive or hazardous materials are used.
- Data Presentation:
- Results: Present the experimental results in tables, figures, or charts.
- Discussion: Interpret the data, draw conclusions, and discuss any observations or unexpected results.
- Documentation and Reporting:
- Lab Notebook: Maintain a detailed lab notebook with records of experimental procedures, observations, and results.
- Report: Prepare a formal report summarizing the experiment, including materials and methods, results, and conclusions.
- Troubleshooting:
- Address any issues or discrepancies that arise during the assay, such as unexpected results or instrument malfunctions.
Safety Considerations in Enzymatic Assays:
Safety considerations are of utmost importance when working with enzymatic assays, especially when handling potentially hazardous materials or using specific techniques. Here are some key safety considerations to keep in mind:
- Personal Protective Equipment (PPE):
- Always wear appropriate PPE, including lab coats, safety goggles, gloves, and closed-toe shoes, to protect against chemical splashes and contamination.
- Chemical Safety:
- Familiarize yourself with the properties of the chemicals used in the assay, including potential hazards and safe handling procedures.
- Use a chemical fume hood when working with volatile or hazardous chemicals to minimize exposure to fumes.
- Label all reagent containers clearly with the name, concentration, and hazard symbols.
- Biological Safety:
- If working with biological samples, follow biosafety protocols and ensure compliance with institutional guidelines for handling potentially infectious materials.
- Dispose of biological waste in designated containers, following appropriate disposal procedures.
- Radiation Safety:
- When using radioactive materials in enzymatic assays, strictly adhere to radiation safety protocols, including shielding, monitoring, and disposal of radioactive waste. Work with radioactive materials in designated areas.
- Equipment Safety:
- Ensure that all laboratory equipment, including spectrophotometers and centrifuges, is in good working condition and regularly maintained to prevent accidents.
- Chemical Compatibility:
- Be aware of chemical compatibility issues, as certain reagents or materials may react unpredictably when combined. Consult safety data sheets (SDS) for compatibility information.
- Emergency Procedures:
- Know the location of safety equipment, such as eyewash stations, safety showers, fire extinguishers, and first-aid kits, and how to use them.
- Familiarize yourself with laboratory evacuation routes and procedures in case of fire, chemical spills, or other emergencies.
- Spill Response:
- Have spill kits readily available and know how to use them. In the event of a chemical spill, follow the appropriate spill response procedures to contain and clean up the spill safely.
- Waste Disposal:
- Dispose of chemical, biological, and radioactive waste in accordance with local regulations and institutional guidelines. Separate waste streams as required.
- Personal Hygiene:
- Wash your hands thoroughly with soap and water after conducting experiments and before leaving the laboratory.
- Avoid eating, drinking, or applying cosmetics in the laboratory to prevent accidental ingestion of hazardous materials.
- Documentation:
- Maintain clear and accurate records of all experimental procedures, including reagent preparations, sample handling, and safety precautions taken.
- Training and Education:
- Ensure that all laboratory personnel, including students and staff, receive appropriate safety training and are aware of the potential hazards associated with enzymatic assays.
- Risk Assessment:
- Conduct a thorough risk assessment before starting an experiment, identifying potential hazards and developing mitigation strategies.
- Supervision:
- Work under the supervision of an experienced researcher or mentor, especially if you are new to enzymatic assays or working with hazardous materials.
- Ethical Considerations:
- Adhere to ethical guidelines and regulations governing the use of animals or human samples in enzymatic assays, ensuring proper informed consent and ethical approval.
Troubleshooting and Common Challenges:
Troubleshooting enzymatic assays is a crucial aspect of experimental work, as various challenges can arise during the setup and execution of these assays. Here are some common challenges and troubleshooting steps to address them:
- Low or Inconsistent Enzyme Activity:
- Solution: Check the enzyme’s activity and stability. Ensure that the enzyme is active, stored correctly, and not expired. Verify that the enzyme is not inhibited by any contaminants in the assay mixture.
- High Background Noise:
- Solution: Use appropriate controls, including negative controls without the enzyme, to identify sources of background noise. Check for contamination of reagents or equipment. Consider using purification or filtration techniques if necessary.
- Poor Assay Sensitivity:
- Solution: Optimize assay conditions such as substrate concentration, pH, and temperature to enhance sensitivity. Adjust the assay duration or increase the enzyme concentration if applicable.
- Nonlinear Kinetics:
- Solution: Ensure that the enzyme reaction is within the linear range by adjusting substrate concentrations. Evaluate whether enzyme inhibition or product inhibition is affecting the reaction.
- Enzyme Inhibition or Activation:
- Solution: Test for potential enzyme inhibitors or activators in the assay mixture. Modify the assay conditions to eliminate or study the effects of these compounds, if necessary.
- High Variability Between Replicates:
- Solution: Pay attention to pipetting accuracy and consistency. Ensure that reagents are well-mixed. Verify the stability of your enzyme and substrates. Increase the number of replicates to improve statistical significance.
- Low Signal-to-Noise Ratio:
- Solution: Increase the signal intensity by using a more sensitive detection method (e.g., fluorometry instead of spectrophotometry). Reduce background noise by optimizing assay conditions and using appropriate controls.
- Instrumental Issues:
- Solution: Calibrate and maintain laboratory instruments regularly. Check for instrument malfunctions or improper settings that may affect data quality.
- Non-Specific Interactions:
- Solution: Use blocking agents or detergents to minimize non-specific interactions between enzymes and surfaces. Ensure that the reaction vessels are clean and properly treated.
- pH and Temperature Fluctuations:
- Solution: Maintain consistent pH and temperature throughout the assay by using proper buffers and temperature control devices. Check for temperature fluctuations in the laboratory environment.
- Sample Contamination:
- Solution: Handle samples with care to prevent contamination. Use sterile techniques for biological samples. Avoid cross-contamination between samples or reagents.
- Low Enzyme Activity in Biological Samples:
- Solution: Optimize sample preparation methods to preserve enzyme activity. Use appropriate extraction or lysis techniques for biological samples, and store them at the recommended conditions.
- Data Analysis Issues:
- Solution: Review data analysis procedures, ensure that calculations are correct, and double-check units and conversion factors. Seek assistance from colleagues or experts if needed.
- Reproducibility Challenges:
- Solution: Document every step of the assay carefully in a laboratory notebook, including reagent preparation, conditions, and any deviations from the protocol. Consult with colleagues or mentors for guidance.
- Consult the Literature and Experts:
- If you encounter persistent issues, consult relevant literature, protocols, or experts in the field for troubleshooting guidance.
Future Trends and Advancements:
The field of enzymatic assays continues to evolve with advancements in technology and an expanding range of applications. Here are some future trends and potential advancements in enzymatic assays:
- High-Throughput Screening (HTS):
- Automation and robotics are becoming increasingly integrated into enzymatic assays, allowing for rapid screening of large compound libraries in drug discovery and functional genomics.
- Microfluidics and Lab-on-a-Chip Technologies:
- Microscale devices and lab-on-a-chip platforms enable miniaturization of assays, reducing sample and reagent volumes while increasing throughput and efficiency.
- Point-of-Care Diagnostics:
- Enzymatic assays are being adapted for use in portable, point-of-care diagnostic devices, enabling rapid and on-site testing for diseases, pathogens, and biomarkers.
- Biosensors:
- Enzyme-based biosensors are advancing for real-time monitoring of specific analytes in applications such as environmental monitoring, healthcare, and food safety.
- Single-Cell Enzymatic Assays:
- Techniques for studying enzyme activity at the single-cell level are emerging, providing insights into cellular heterogeneity and rare cell populations.
- Multi-Omics Integration:
- Enzymatic assays are being integrated with genomics, transcriptomics, and proteomics data to provide a more comprehensive understanding of cellular processes and diseases.
- Advanced Imaging Techniques:
- Imaging technologies are improving the visualization of enzyme activity within cells and tissues, allowing for the study of spatial and temporal dynamics.
- AI and Machine Learning:
- AI-driven approaches are being used to analyze complex enzymatic assay data, identify patterns, and predict enzyme behavior, aiding in drug discovery and enzyme engineering.
- Crispr-Cas Enzyme Assays:
- Enzymatic assays are being developed to characterize CRISPR-Cas enzymes and their variants, advancing genome editing research and applications.
- 3D Culture Systems:
- Enzymatic assays in three-dimensional (3D) cell culture systems are providing more physiologically relevant insights into enzyme function and drug response.
- Metabolomics and Enzyme Profiling:
- Metabolomic approaches, combined with enzymatic assays, are uncovering novel metabolic pathways and biomarkers associated with diseases and drug responses.
- Green and Sustainable Assays:
- The development of environmentally friendly and sustainable enzymatic assays, using eco-friendly substrates and enzymes, is gaining importance.
- Synthetic Biology and Directed Evolution:
- Synthetic biology tools and directed evolution techniques are being employed to engineer enzymes with enhanced properties for specific applications.
- In Silico Assay Design:
- Computational modeling and in silico simulations are being used to design enzymatic assays, predict enzyme-substrate interactions, and optimize assay conditions.
- Personalized Medicine:
- Enzymatic assays are contributing to the advancement of personalized medicine by enabling the identification of patient-specific biomarkers and drug responses.
FAQs:
What is an enzyme?
Enzymes are biological molecules that act as catalysts, speeding up chemical reactions in living organisms.
What are enzymatic assays?
Enzymatic assays are laboratory techniques used to measure the activity of enzymes or quantify specific substrates, products, or coenzymes in biological samples.
Why are enzymatic assays important?
Enzymatic assays are crucial for understanding enzyme function, drug development, clinical diagnostics, and various industrial applications.
What are the types of enzymatic assays?
Types include spectrophotometric, fluorometric, radioactive, colorimetric assays, and more, each with specific applications.
What are the principles of enzymatic assays?
Principles include specificity, enzyme-substrate interaction, reaction kinetics, linearity, and standardization, among others.
What are common enzymatic assay techniques?
Common techniques include spectrophotometry, fluorometry, chromatography, ELISA, and enzyme activity assays.
What are specialized enzymatic assay methods?
Specialized methods target specific enzymes or biochemical processes, such as kinase assays or glycosidase assays.
What safety considerations apply to enzymatic assays?
Safety considerations include wearing PPE, handling chemicals and biological materials safely, and following safety protocols for hazardous materials.
What are some troubleshooting tips for enzymatic assays?
Troubleshooting involves addressing issues like low enzyme activity, high background noise, or poor assay sensitivity through systematic investigation.
What are the future trends in enzymatic assays?
Trends include high-throughput screening, microfluidics, point-of-care diagnostics, biosensors, AI, and more.
What is the Michaelis-Menten equation?
The Michaelis-Menten equation describes the relationship between substrate concentration and enzyme activity, helping determine Km and Vmax values.
What is ELISA used for?
ELISA (Enzyme-Linked Immunosorbent Assay) is used for detecting and quantifying specific antigens or antibodies, commonly used in clinical diagnostics and immunology.
How do you measure enzyme kinetics?
Enzyme kinetics are measured by studying the rate of product formation or substrate consumption under varying conditions and plotting the data for analysis.
What is the Lineweaver-Burk plot?
The Lineweaver-Burk plot is a graphical representation of enzyme kinetics data, used to determine kinetic parameters like Km and Vmax.
What are biosensors?
Biosensors are devices that incorporate biological components, including enzymes, to detect specific analytes, often in real-time.
What is the role of enzymes in molecular biology?
Enzymes in molecular biology, like DNA polymerases and nucleases, are used in DNA replication, PCR, and gene expression studies.
What are the applications of enzymatic assays in drug discovery?
Enzymatic assays are used to screen and evaluate potential drug candidates, identify enzyme inhibitors, and study enzyme-drug interactions.
How can I ensure reproducibility in enzymatic assays?
Reproducibility is ensured by using standardized protocols, controls, proper documentation, and regular calibration of instruments.
What are the benefits of 3D culture systems in enzymatic assays?
3D culture systems provide a more physiologically relevant environment for studying enzyme function and drug responses in cell-based assays.
Conclusion:
In conclusion, enzymatic assays are foundational tools in the fields of biology, biochemistry, and beyond. Their significance lies not only in their ability to elucidate the intricate workings of enzymes and biochemical pathways but also in their diverse applications, from advancing drug discovery to revolutionizing clinical diagnostics. As technology continues to advance and our understanding of enzymatic processes deepens, enzymatic assays will remain indispensable, paving the way for breakthroughs in science, medicine, and industry, and addressing the ever-evolving challenges of our complex biological world.