Enzyme-Linked Immunosorbent Assay (ELISA)
The definitive clinical and research guide mapping plate-based methodologies, solid-phase specificity, robust quantification protocols, and advanced troubleshooting.
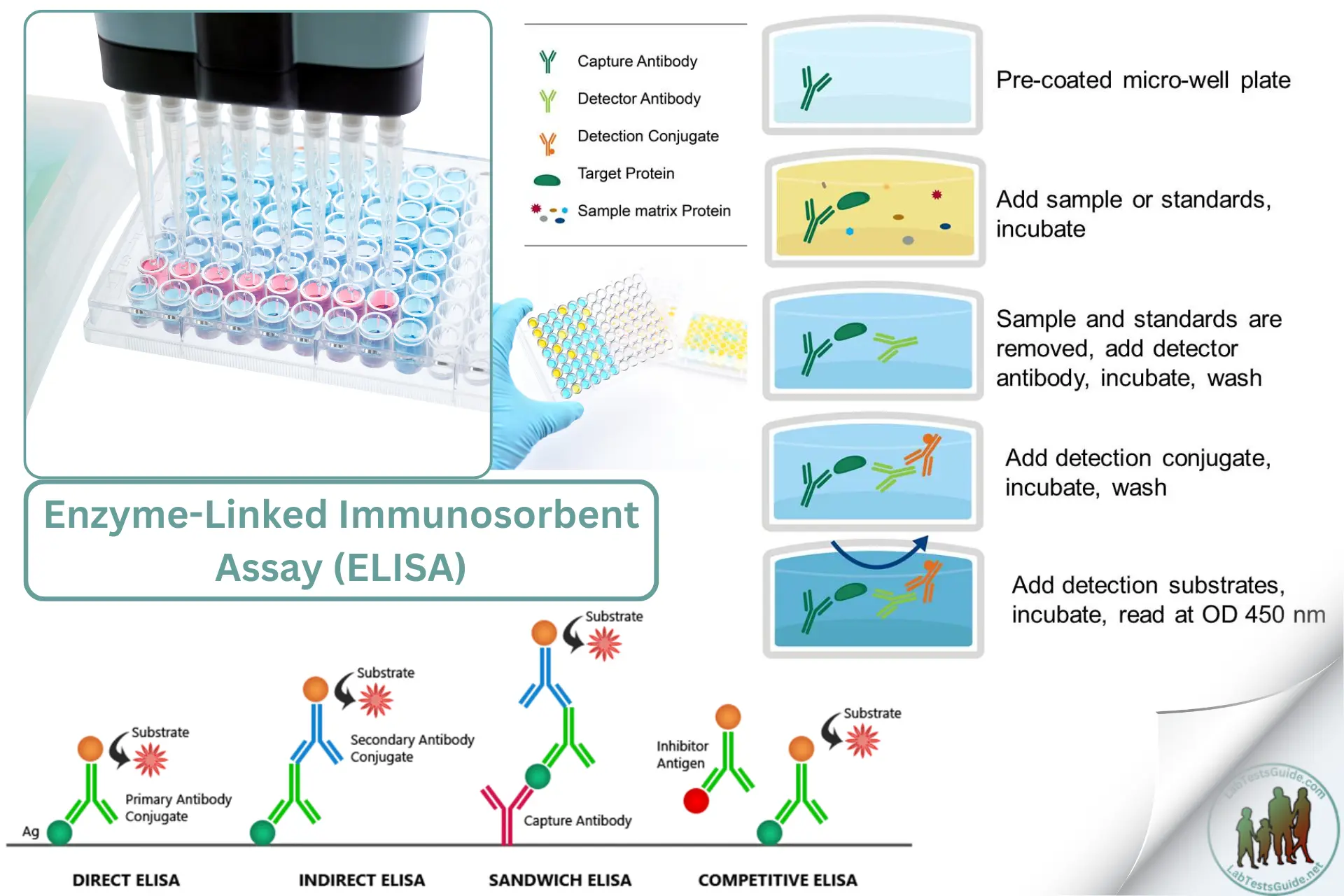
Introduction to ELISA
Definition: ELISA is a highly sensitive, plate-based assay technique designed for detecting and quantifying soluble substances such as peptides, proteins, antibodies, and hormones. It is a specific, heterogeneous subset of EIA.
Overview: What defines ELISA is the Immunosorbent phase. Unlike generalized fluid immunoassays, ELISA requires the target antigen or antibody to be physically immobilized onto a solid surface (typically a 96-well polystyrene microplate). This allows for rigorous washing steps, vastly improving the signal-to-noise ratio.
Historical Context: Created in 1971 by Peter Perlmann and Eva Engvall at Stockholm University. Their goal was to find a highly sensitive, quantitative replacement for Radioimmunoassay (RIA) that didn’t expose lab technicians to hazardous radiation.
Modern Diagnostic Importance: Today, ELISA is the global gold standard in clinical pathology for infectious disease screening (like HIV and Syphilis), allergy testing, and biomarker quantification in biomedical research.
Fundamental Principle
The Solid Phase (Immobilization)
Antigens or antibodies are firmly attached to the plastic surface of microplate wells via hydrophobic interactions. This allows subsequent reagents to be added and washed away without losing the bound target.
Lock-and-Key Specificity
The system utilizes the extreme biological specificity of an antibody seeking its exact target antigen, preventing false cross-reactivity with other proteins in the patient’s serum.
Enzymatic Amplification
Once bound, a detection antibody linked to an enzyme (like HRP) is introduced. The enzyme rapidly converts a colorless substrate (like TMB) into a measurable colored product.
Quantitative Absorbance
A spectrophotometer (Microplate Reader) shoots specific wavelengths of light through the well. The Optical Density (OD) recorded is directly proportional to the amount of target present.
The Four Primary ELISA Formats
The structural format of the assay dictates its sensitivity, specificity, and exact clinical application.
Direct ELISA
The antigen is directly coated to the plate. A conjugated primary antibody binds it directly. Pros: Fast protocol. Cons: Lower sensitivity due to lack of signal amplification.
Indirect ELISA
Antigen is coated. An unlabeled primary antibody binds it, followed by an enzyme-labeled secondary antibody. Pros: High sensitivity; ideal for testing patient antibody levels.
Sandwich ELISA
A “Capture” antibody is coated. Sample antigen binds, then a “Detection” antibody binds the antigen (creating a sandwich). Pros: The most powerful, highly specific format.
Competitive ELISA
Sample antigen and an enzyme-labeled reference antigen compete to bind a limited number of coated antibodies. Pros: Excellent for detecting very small molecules (e.g., hormones, drugs).
Pre-Analytical Guidelines & Specimen Prep
Sample Collection
- Serum & Plasma: The standard for clinical tests. Draw in SST or EDTA tubes. Centrifuge to separate.
- Cell Culture Supernatants: Centrifuge at 1000 x g to remove cellular debris before plating.
- Tissue Homogenates: Requires strict lysis buffers supplemented with protease inhibitors to protect target proteins from degradation.
Handling & Storage Stability
- Storage: Centrifuged serum should be kept at 2-8°C if assayed within 5 days. For long-term archiving, freeze at -20°C to -80°C.
- Caution: Strictly avoid repeated freeze-thaw cycles, as this degrades tertiary protein structures, destroying antibody binding sites and causing false negatives.
Clinical ELISA Protocol & Laboratory Method
This is the standard, optimized laboratory procedure utilized in clinical diagnostics and research biology. Exact incubation times and volumes may vary slightly by manufacturer kit, but adhere strictly to these fundamental mechanics.
Coating the Microplate
Dilute the capture antigen or antibody in a high-pH Coating Buffer (usually Bicarbonate/Carbonate buffer, pH 9.6). Dispense the fluid into the 96-well polystyrene plate. Seal the plate to prevent evaporation.
Plate Washing (Post-Coat)
Discard the coating solution. Vigorously wash the plate to remove unattached proteins. Invert the plate and blot dry against clean paper towels to ensure no residual buffer remains.
Blocking Unbound Sites
Add Blocking Buffer to saturate any remaining empty plastic surface area in the wells. This is a critical step to prevent non-specific binding of future reagents, which causes high background noise.
Sample & Standard Incubation
Wash the plate 3 times. Add the prepared patient samples, assay controls, and standard curve dilutions to specific designated wells. During this phase, the target analyte selectively binds to the coated capture proteins.
Enzyme Conjugate Addition
Add the detection antibody conjugated with an enzyme (typically Horseradish Peroxidase – HRP, or Alkaline Phosphatase – ALP). This conjugate binds specifically to the target analyte currently trapped in the well.
Substrate Addition (Color Development)
Add the chromogenic substrate. For HRP, TMB is utilized. The enzyme catalyzes the substrate, turning the clear liquid into a vibrant blue color. This step is light-sensitive and must be performed in the dark.
Stopping the Reaction & Reading
Add an acidic Stop Solution to rapidly denature the enzyme and halt color development. The solution instantly changes from blue to yellow. Immediately insert the plate into the Microplate Spectrophotometer to read the Optical Density (OD).
Reagents & Catalytic Substrates
| Component Category | Specific Examples | Clinical Function |
|---|---|---|
| Microplates | 96-well Polystyrene | High protein-binding capacity solid phase for immobilization. |
| Wash Buffers | PBS-T, TBS-T (Tween-20) | Surfactants that disrupt weak, non-specific molecular bonds. |
| Enzyme Conjugates | HRP, Alkaline Phosphatase | The catalytic engine that drives signal amplification. |
| Substrates | TMB, OPD, PNPP | Chemicals converted by the enzyme into measurable optical colors. |
Quantitative Interpolation Engine
In clinical quantitative ELISA, a Standard Curve is generated using known concentrations. Patient sample optical densities are then interpolated against this curve to find exact analyte concentrations in the blood.
Clinical & Research Applications
Infectious Pathology
- Viral: HIV p24 antigen and antibodies,Anti HCV, Hepatitis B Surface Antigen (HBsAg), SARS-CoV-2, etc.
- Bacterial: Syphilis, Lyme Disease, H. pylori, etc.
Endocrinology & Oncology
- Hormones: Thyroid (TSH, T4), Reproductive (hCG, FSH, LH).
- Tumor Markers: PSA (Prostate), CEA, CA-125, etc.
Immunology
- Autoimmune: ANA (Anti-nuclear antibodies), Rheumatoid Factor, etc.
- Allergy: Quantification of specific IgE antibodies in serum, etc.
Biomedical Research
- Cytokine Profiling: Measuring interleukins to track inflammation.
- Vaccine Development: Titering neutralizing antibodies post-immunization.
Assay Architecture: Strengths, Limitations & Troubleshooting
Primary Advantages
- Exquisite sensitivity (detecting pg/mL ranges).
- Easily automated for 96 or 384-well high-throughput.
- Highly reproducible with robust shelf-life reagents.
Core Limitations
- Dependent on highly specific antibody availability.
- “Hook Effect” can cause false negatives in vastly concentrated samples.
- Lengthy manual incubation periods compared to rapid lateral flow.
Advanced Troubleshooting Matrix
Issue: High Background Noise (False Positives)
Occurs when the entire plate turns slightly blue. Causes: Inadequate blocking buffer concentration, insufficient washing technique, or severe cross-contamination between wells. Ensure washer pins are unclogged.
Issue: Weak or Zero Signal
Standard curve fails to develop color. Causes: Omission of a critical reagent (like the conjugate), enzyme degradation due to poor storage, presence of sodium azide (which kills HRP), or expired substrates.
Issue: Edge Effect & High CV%
Wells on the outside edge of the plate show higher ODs than the center. Causes: Evaporation during incubation or uneven thermal gradients. Always use plate sealers and ensure the incubator is fully pre-heated.
Appendices & Future Horizons
The Future of ELISA
The transition toward Multiplexing allows the detection of up to 50 distinct cytokines in a single well utilizing encoded microbeads. Furthermore, Digital ELISA utilizing microwell arrays is pushing boundaries, allowing the counting of single enzyme molecules, lowering detection limits by 1000x.
Frequently Asked Questions (FAQs)
Why use 96-well plates? It is the global standard format, optimizing reagent volume (usually 100µL per well) and perfectly fitting automated readers.
What is the “Hook Effect”? In a sandwich ELISA, an extraordinarily high concentration of patient antigen can saturate both capture and detection antibodies separately, preventing the “sandwich” from forming, falsely resulting in a low reading.
How long does an ELISA take? A standard laboratory protocol takes 3 to 4 hours from sample addition to reading.
Glossary of Terms
Immunosorbent: The ability of a solid surface (plastic plate) to absorb and hold immune proteins.
TMB (Tetramethylbenzidine): The most common chromogenic substrate used with HRP, turning blue when oxidized.
Standard Curve: A graph created by plotting ODs of known concentrations, utilized to calculate unknown patient samples.
Wash Buffer: A saline solution with detergents used to sweep away unreacted proteins.
References:
- Engvall, E., & Perlmann, P. (1971). Enzyme-linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry, 8(9), 871-874.
- Tietz Textbook of Clinical Chemistry and Molecular Diagnostics
- Manufacturer’s instructions for ELISA reagent kits
- Clinical Laboratory Standards Institute (CLSI) guidelines







